Patient and Public Involvement and Engagement Strategy
Acronyms
We have tried to minimise acronyms in this strategy. The ones we have used can be found below:
BRC: Biomedical Research Centre
CRF: Clinical Research Facility
EDI: Equality, Diversity, and Inclusion
KCH: King’s College Hospital
KCL: King’s College London
NIHR: National Institute for Health and Care Research
PPI/E: Patient and Public Involvement/Engagement
SLaM: South London and the Maudsley NHS Foundation Trust
At the end of each section of the strategy there will be a blue hyperlink which will bring you back to this ‘acronyms’ section where you can view the acronyms written out in full.
Definitions
Key definitions from the National Institute for Health and Care Research (NIHR) are listed:
Clinical Research Facility: purpose-built facilities in NHS hospitals where researchers can deliver studies. These studies may be in an early phase (when a researcher is looking at whether something is safe or effective).
Clinical Trial: an experiment to compare the effects of two or more medicines, treatments, or procedures. ‘Clinical trial’ is an umbrella term for a variety of different experiments using different methods.
Engagement: where information and knowledge about research is provided and disseminated to the general public, for example via science festivals, open days, and media. coverage.
Impact: the changes, benefits, and learning gained from the insights and experiences of patients, carers, and the public when working in partnership with researchers and others.
Involvement: an active partnership between public members and researchers in the research process, rather than the use of people as “subjects” or “participants” of research. It is often defined as doing research “with” or “by” members of the public rather than “to”, “about” or “for them”.
Participation: the act of taking part in a research study, for example being recruited to take part in a clinical trial.
At the King’s Clinical Research Facility our participants may be local (from the Southeast London area) or may travel from further away (from across the country or abroad).
Public members: include patients, potential patients, carers, and people who use health and social care services as well as people from organisations that represent people who use services.
Research: discovering new knowledge that could lead to changes in treatments, policies, or care.
Introduction
This strategy outlines our ambition, aim, principles, and goals for Patient and Public Involvement and Engagement (PPIE) at the King’s Clinical Research Facility (CRF).
We want to build meaningful and respectful PPIE, which improves the relevance, quality, and impact of the research and work which takes place here.
Our work at the King’s Clinical Research Facility is funded substantially by the NIHR, and we use their definitions of involvement and engagement in research. Involvement in research is when research is carried out “with” or “by” members of the public rather than “to”, “about” or “for them”. Engagement, on the other hand, is where information and knowledge about research is communicated.
Involvement and engagement of members of the public is essential. It helps us to understand the needs, concerns, and priorities for research of those who use health and social care services.
We want staff, researchers, and public members to read and use this document often. We understand that involvement and engagement can be challenging. It takes time to build relationships and trust.
The King’s Clinical Research Facility
The King’s Clinical Research Facility is designed to support clinical trials on a broad range of topics including mental health and general medicine. These clinical trials may be sponsored by pharmaceutical companies (known as commercial trials) or sponsored by the NHS, Research Councils or charities (known as non-commercial trials).
The King’s Clinical Research Facility is made up of four research areas listed below. They are all based physically in King’s College Hospital acting in partnership with South London and the Maudsley NHS Foundation Trust and King’s College Hospital.
- The Experimental Medicine Facility: this contains rooms set up with clinical equipment, such as the equipment needed to take blood or to give medication, and specialised rooms where volunteers and patients with particular conditions taking part in clinical trials can be interviewed.
- The Cell Therapy Unit: where human-cell and gene-based therapies can be produced.
- The Clinical Trials Facility: this contains ward beds and rooms where volunteers and patients with particular conditions taking part in clinical trials can be examined.
- The Imaging Facility: this contains a 3T magnetic resonance imaging scanner, and we are expecting a second scanner to be installed in 2023.
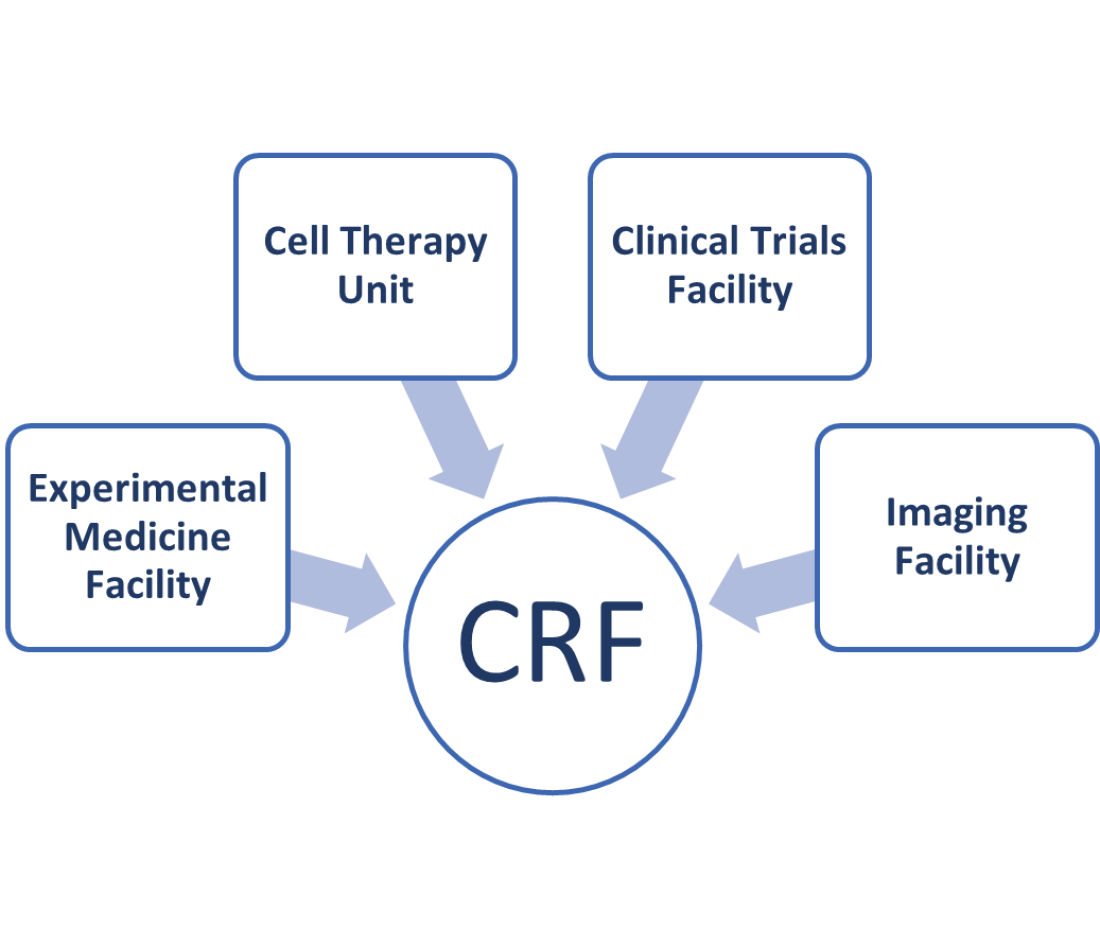
Research areas within the King’s Clinical Research Facility
The King’s Clinical Research Facility has clinical research and support staff to help research teams with their studies. Research teams from across the hospital undertaking commercial or non-commercial trials must apply to use the CRF and to work with our staff.
The wider King’s Clinical Research Facility Strategy outlines our aim to provide an excellent facility for the efficient and safe conduct of clinical trials. We hope these clinical trials will lead to developments that transform the lives of patients. We believe this aim can only be achieved by working alongside public members who have:
- Experience of what it is like to be part of a clinical trial
- Experience of living with/or caring for someone living with a certain health condition
- An interest in clinical trials, and the health conditions frequently studied at the King’s Clinical Research Facility
Our partners and collaborators
We receive support from many places. Our partners and collaborators have influenced this Patient and Public Involvement and Engagement Strategy as follows:
King’s Health Partners: We receive academic support from King’s Health Partners [1]. King’s Health Partners is a collaboration between hospitals and universities in the Southeast London area. A guiding principle for King’s Health Partners is to put members of the public first by ensuring they are at the heart of everything. This Public Involvement and Engagement Strategy supports King’s Health Partners’ ethos.
King’s College Hospital: we are based in King’s College Hospital, and therefore we align ourselves with the hospital strategy, which is to be BOLD [2]:
- Consisting of Brilliant people
- Delivering Outstanding care
- Developing Leaders in research and education
- With Diversity, Equality, and Inclusion at the centre of it all
Representatives from the King’s College Hospital Patient Experience and Engagement Team have provided us with PPIE support and guidance.
- King’s College London: we are also based in King’s College London. We plan to collaborate with already established PPIE teams such as the King’s College London Impact and Engagement Services Team and the King’s College London PPI Lead for the Centre of Rheumatic Diseases.
- South London and the Maudsley (SLaM) and its Biomedical Research Centre (BRC): We have significant interactions with SLaM and its BRC. The SLaM Service User Advisory Group have, and will continue to, provide us with PPIE support and guidance.
- NIHR: we receive funding from the NIHR. We are also supported by the NIHR’s PPIE infrastructure, available resources, and guidance. The NIHR Applied Research Collaboration (ARC) South London hosts events and provides training and support in PPIE.
To note: We also received funding from the Wellcome Trust when we were first established.
- Public members: our public members are key partners and collaborators. As more public members join us, their local community contacts and networks will be invaluable in helping us to build a diverse group. We want the group to be reflective of the South London population we serve.
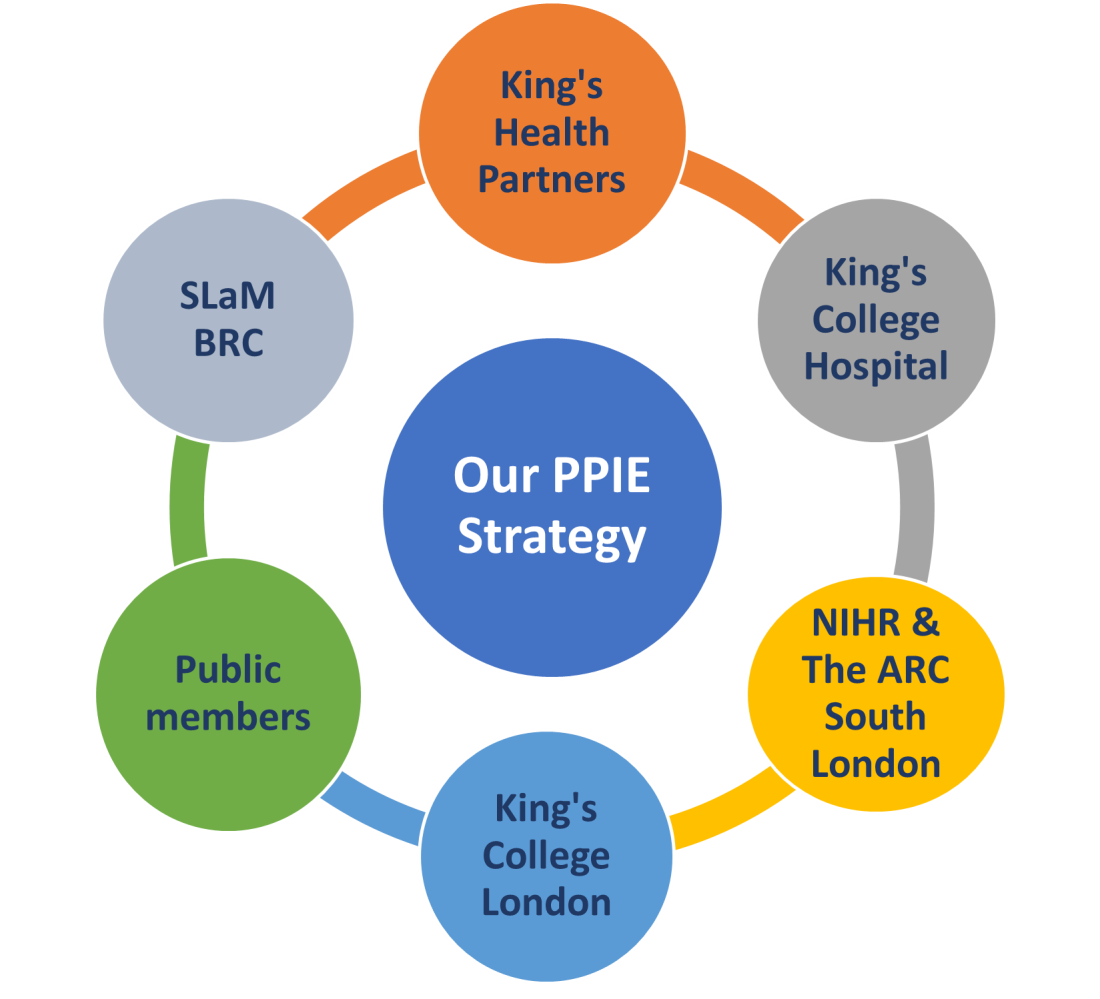
Partners and collaborators that have influenced our strategy
Our ambition and aim for PPIE
Our ambition: to develop a culture that places PPIE at the heart of all aspects of our work.
Our aim: to raise awareness of our research, and to promote meaningful and attractive PPIE opportunities related to our research and work.
Our ambition and aim are built on and in line with the NIHR’s PPIE Strategy, ‘Going the Extra Mile’ [1]. Their strategy outlines the need for the public as partners in all work that is undertaken, and the need for involvement and engagement opportunities to be visible and seized by the public.
Our principles
- Responsive patient and public involvement: we will take ideas onboard, and we will make changes when they are needed
- Respectful patient and public involvement: we will take time to listen to, and learn from, diverse opinions and voices. We will do this with the same level of enthusiasm and interest. We must create an environment where the learning is two-way (between public members and staff members)
- Relationship-centred involvement: we will value everyone and the unique experiences they bring to our work
Our principles were developed drawing on the approaches and behaviours encouraged by the UK Standards for Public Involvement [1]. Whilst all six standards are aspirational, we particularly focused on working together (working in a way that values all contributions) and communication (using clear language for well-timed and relevant communication). See Appendix 1.
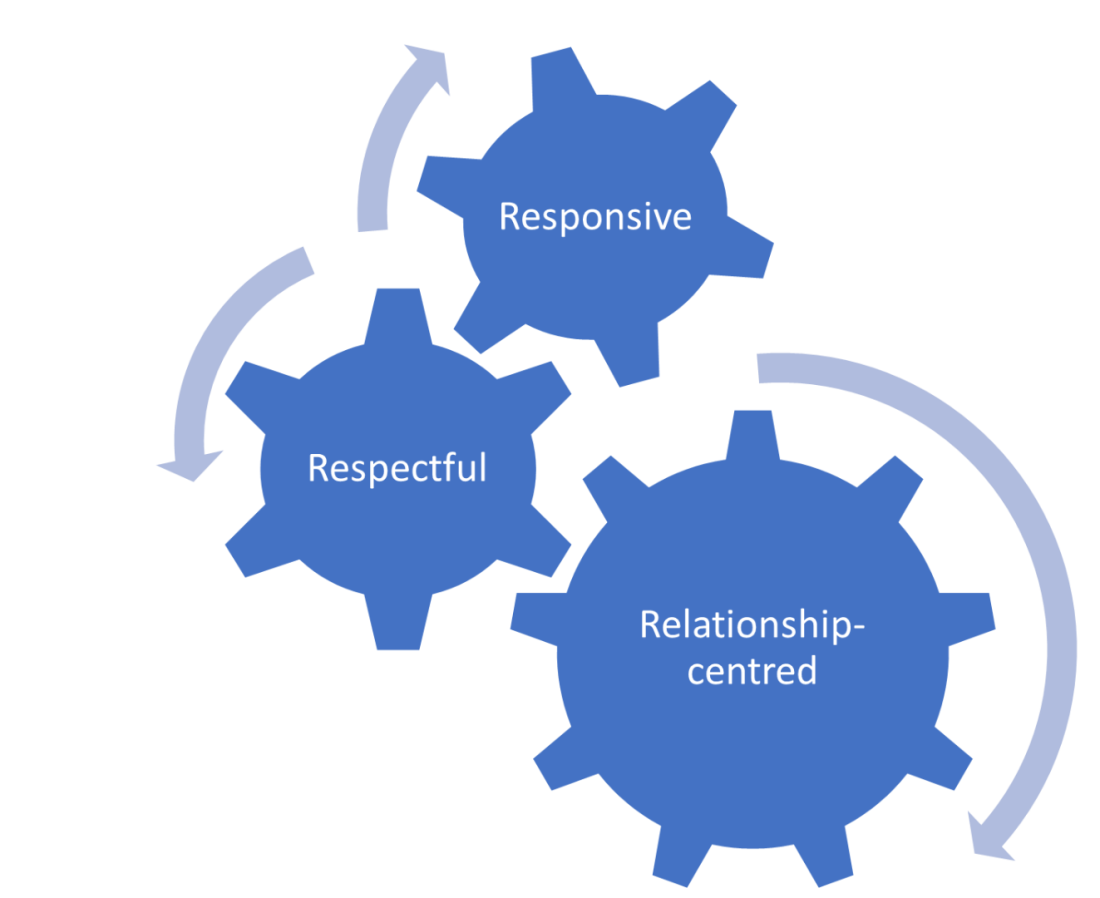
Our principles
Our goals
Goal 1: To promote the King’s Clinical Research Facility, our research and work in a way that attracts members of the public (to join our public involvement group, and to engage with relevant events)
Goal 2: Offer a first-class experience for members of the public who visit the King’s Clinical Research Facility and are involved in our work
Goal 3: To engage and educate young people and academic institutions on the work of the King’s Clinical Research Facility and the importance of PPIE
Goal 4: Evaluate the impact of public involvement and engagement in our research
Our action plan
For goal 1, we plan to:
- Co-create content for the PPIE webpage with public members. The webpage will be used to share information and reach a wider population of public members and researchers. We will do this by mid 2023, then update content when needed. (Responsible individuals: PPIE Lead and Communications Officer)
- Use social media platforms, such as the King’s Clinical Research Facility Twitter account, to share information and promote engagement activities with a wider population of public members and researchers. We will do this by early 2023, then update content when needed. (Responsible individuals: Communications Officer)
- Install electronic, interactive noticeboards in the CRF to display PPIE information (e.g., involvement opportunities and upcoming events) and other helpful information about the CRF including ‘currently recruiting’, and relevant Twitter feeds. We will do this by mid 2023, then update content quarterly. (Responsible individuals: PPIE Lead)
- Revise and update the welcome pack for research participants who attend the CRF. Co-create a welcome pack with public members for new members as they join us. We will do this by the end of 2023. (Responsible individuals: PPIE Lead, Communications Officer, and CRF Manager)
- Develop a bi-annual newsletter that participants and public involvement members can sign up to for information about relevant CRF news and upcoming events. We will do this by mid 2023, then distribute the newsletter bi-annually. (Responsible individuals: PPIE Lead and Communications Officer)
- Identify opportunities to learn from patient awareness groups, promote the work of the CRF, and join patient awareness-specific health days (e.g., Mental Health Day, Migraine, Diabetes, Alzheimer’s, Depression). We will do this by mid 2023, then annually. (Responsible individuals: PPIE Lead and Communications Officer)
- Identify opportunities to advertise throughout the trust including to KCH, SLaM, and KCL. We will do this by mid 2023. (Responsible individuals: PPIE Lead and Communications Officer)
For goal 2, we plan to:
- Develop a Strategy Group composed of 8-10 core public members and CRF staff to oversee putting this strategy into practice and to make changes where needed. Advert for Strategy Group members to be placed in People in Research [1] with informal virtual interviews to be held to select individuals with diverse backgrounds and experiences. See Appendix 2. To be co-chaired by a public member and CRF staff member. We will do this by mid 2023, then meet quarterly. (Responsible Individuals: PPIE Lead and CRF Manager)
- Introduce a training needs survey to understand what training is required for public members and CRF staff to ongoingly improve everyone’s experience of PPIE. We will do this by end of 2023. (Responsible Individuals: PPIE Lead and CRF Manager)
- Develop training on PPIE for CRF staff and researchers to be co-delivered with a public member during regular Friday teaching sessions (x 2 per year). All staff must attend at least 1 of the sessions. We will do this by early 2024, then training will be delivered bi-annually. (Responsible Individuals: PPIE Lead)
- Co-develop and co-deliver induction sessions for PPIE members and ‘knowledge exchange’ events for PPIE members and researchers with the PPIE Lead for the CRF at Guy’s and St. Thomas’. ‘Knowledge exchange’ events to include sharing best practice in involvement and engagement. We will do this by early 2024. (Responsible Individual: PPIE Lead)
- Identify further members to join the public involvement group and put together a secure contact database of interested parties. This will ensure we have a wide range of stakeholders to extend the reach of our communications and involvement and engagement activities. We will do this by end of 2023, then ongoingly. (Responsible Individuals: PPIE Lead)
- Allocate public members to specific research projects in their areas of interest or expertise (e.g., mental health, haematology). We will do this by early 2024, then ongoingly. (Responsible individual: PPIE Lead)
- Develop ‘dragon’s den’ events where public members can give immediate input on researchers’ work (including ideas for recruitment, dissemination, etc.). We will do this by mid 2024, then bi-annually. (Responsible individual: PPIE Lead)
- Develop an online community for the public involvement group. This may include opportunities for informal support, empowerment, and networking. We will do this by early 2025. (Responsible individuals: PPIE Lead, Communications Officer, and Data Manager)
For goal 3, we plan to:
- Continue to work with local schools and offer work experience and/or open days for GCSE and A-level students. (Responsible individuals: PPIE Lead and CRF Manager)
- Continue to work with Project Search [2] to offer work experience for young adults with learning disabilities and autism or both. We will do this by mid 2023, then annually. (Responsible individuals: PPIE Lead, Research Nurses, and CRF Manager)
For goal 4, we plan to:
- Develop feedback mechanisms, such as a short survey to ascertain how many people know about the CRF, if they would be interested in participating in research, and if they would like to join the PPI We will do this annually. (Responsible individuals: PPIE Lead and CRF Manager)
- Continue to use the ‘data at the door’ survey with useful data about visitors to the CRF at King’s College Hospital. We will do this annually. (Responsible individuals: PPIE Lead and CRF Manager)
- Monitor and reflect on progress with specific public involvement activities and events using evaluation forms, which are co-produced with public members. We will do this annually. (Responsible individuals: PPIE Lead and CRF Manager)
- Towards the end of the strategy invite public members to take part in focus groups or interviews to discuss progress made against each goal and their experience of being involved in the strategy group. We will do this by mid 2025. (Responsible individuals: PPIE Lead and CRF Manager)
Our roadmap
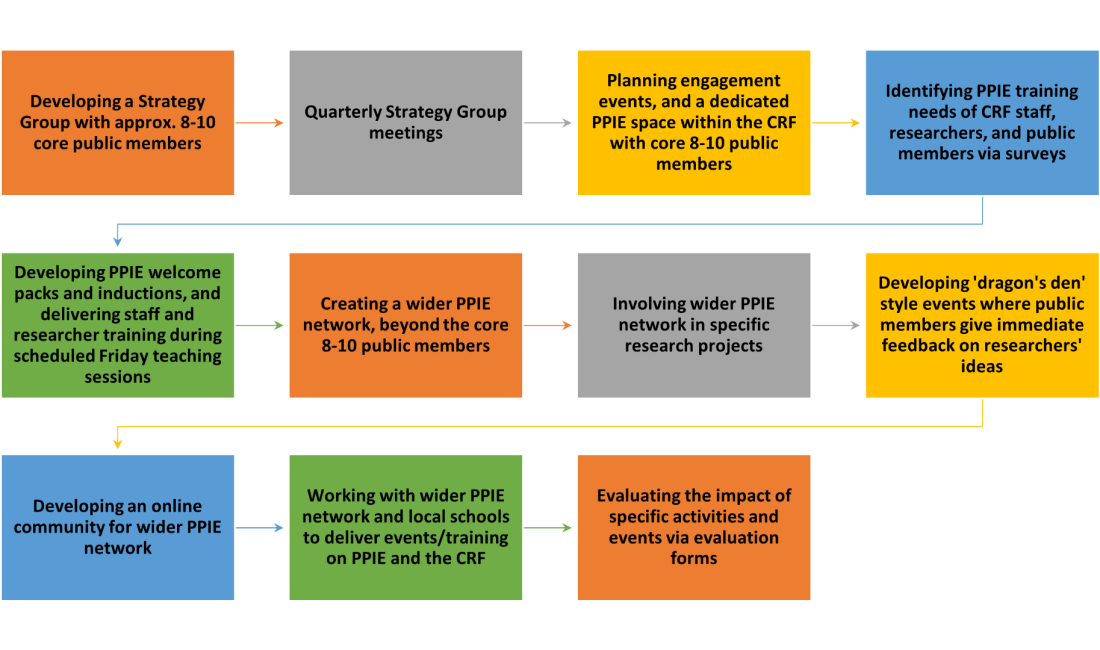
Resources and infrastructure supporting our strategy
Our strategy development and delivery will be supported by our:
CRF Manager – with overall responsibility for putting this strategy into practice
PPIE Lead – with responsibility for ensuring effective involvement throughout the CRF’s activities
Our partners and collaborators at King’s College Hospital, King’s College London, South London and the Maudsley and its Biomedical Research Centre, the NIHR, and our public members will also provide support and guidance in the PPIE work undertaken and outlined in this strategy.
There are funds to support public involvement activity. Public members will be paid for their time at a rate of up to £25 per hour, in line with current NIHR Centre for Engagement and Dissemination benchmarks [1]. Public members’ reasonable travel expenses will also be reimbursed. We want to make sure that there is equality of opportunity, and that payment is not a barrier for any public members who wish to be involved in the work and activities of the King’s Clinical Research Facility.
Whilst this strategy does not cover other issues related to Equality, Diversity, and Inclusion (EDI) in depth, it is an important aspect of any public involvement work. Alongside offering payment, we will be mindful of individuals’ backgrounds and range of experiences and interests when joining us to ensure as diverse a public involvement group as possible. We will also offer any welcome, induction or training materials in accessible formats (such as large print) as required.
As part of this strategy, we are also proposing the following key structures to support involvement at the King’s Clinical Research Facility:
– Strategy Group: a small group made up of public members and staff members will review progress made against the strategy goals every quarter as outlined in goal 2. Members will also help the CRF Manager and PPIE lead to explore how involvement might be strengthened more generally.
– An active learning series: the PPIE lead will co-deliver informal training with a public member during established Friday teaching sessions (x 2 per year). The training will be for staff about what public involvement is, and how they best can support involvement and engagement activities as outlined in goal 1. The PPIE lead will also develop a welcome pack for new public members as outlined in goal 1.
[1] https://www.nihr.ac.uk/documents/payment-guidance-for-researchers-and-professionals/27392
Our governance structures
Our Strategy Group will ensure that public members are integral in the governance of the King’s Clinical Research Facility, and that their ideas and views are actively sought and taken up by staff. We will also:
- Add PPIE, and any EDI considerations as a standing agenda item in monthly management board meetings.
- Invite up to 3 public members to sit on our governance committee. The public members may meet with a sub-group of the governance committee to feedback ideas, and to report on PPIE activities. We want to make sure that PPIE members feel their presence is valued, and that they are supported to contribute to governance committees in a way that works for everyone.
- Designate up to 4 involvement champions among the CRF research nurses and clinical research practitioners. The involvement champions will be an additional point of contact and support for public members who wish to be involved. They will also encourage good practice and distribute PPIE documents and guidance to public members and other staff. Involvement champions may be designated to attend PPIE meetings or events in the place of the PPIE Lead if she is unavailable.
For reporting purposes, the PPIE Lead is accountable to Elka Giemza, CRF Manager. The CRF Manager is accountable to Professor Peter Goadsby, the Director of the CRF.
Evaluating our strategy
We will:
- Review progress with our strategy during quarterly meetings (with the Strategy Group once this has been developed, and before this with the CRF Manager and other relevant staff).
- Gather evaluation forms and ongoing feedback on public involvement activities and events as part of goal 4.
- Towards the end of 2025, obtain feedback via a survey from CRF staff, researchers, research participants, and public members about progress made against each goal. This will inform the next 3 years (2025-2028) of the strategy.
- Towards the end of 2025, use the UK Standards as an evaluation tool [1] to identify areas of strength and areas for improvement in our public involvement and engagement. This will inform the next 3 years (2025-2028) of the strategy.
- Towards the end of 2025, invite public members to take part in focus groups or interviews to discuss progress made against each goal and their experience of being involved in the strategy group. This will inform the next 3 years (2025-2028) of the strategy.
- Use the Public Involvement in Research Impact (PIRI) Toolkit developed by the Marie Curie Research Centre and the Wales Cancer Research Centre [2], to record the contributions public members have made, and the changes to research and practice as a result. See Appendix 3.
[1] Seddon, K., Elliott, J., Johnson, M. et al. Using the United Kingdom standards for public involvement to evaluate the impact of public involvement in a multinational clinical study. Res Involv Engagem 7, 22 (2021). https://doi.org/10.1186/s40900-021-00264-3
Acknowledgements
We would like to thank the following individuals who reviewed and improved this strategy:
- The 10 members of the King’s Clinical Research Facility PPI Group
- Professor Peter Goadsby, Director, King’s Clinical Research Facility
Contact
To find out more or to get involved with public involvement and engagement at the King’s Clinical Research Facility, please email: flora.siklosi@nhs.net
Appendix 1. The UK Standards for Public Involvement
UK Standards for Public Involvement
Inclusive opportunities
Offer public involvement opportunities that are accessible and that reach people and groups according to research needs.
Working together
Work together in a way that values all contributions, and that builds and sustains mutually respectful and productive relationships.
Support and learning
Offer and promote support and learning opportunities that build confidence and skills for public involvement in research.
Governance
Involve the public in research management, regulation, leadership and decision making.
Communications
Use plain language for well-timed and relevant communications, as part of involvement plans and activities.
Impact
Seek improvement by identifying and sharing the difference that public involvement makes to research
Appendix 2. Advert in People in Research


Appendix 3. Public Involvement in Research Impact Toolkit
Public Involvement in Research Impact Toolkit planning tool snapshot:

The Public Involvement in Research Impact Toolkit (PIRIT) was developed by the Marie Curie Research Centre and the Wales Cancer Research Centre.
